Developmental Neuropathology
Chapter 7: Cerebral heterotopia
Heterotopion: malformative lesion comprised of normal cells in the wrong place. Three types:
- leptomeningeal heterotopia - focal or diffuse collections of glioneuronal tissue within the leptomeninges
- also called "leptomeningeal glioneuronal heterotopia" or "meningeal heterotopia"
- contiguous with underlying marginal zone or brain parenchyma
- most commonly multiple and associated with numerous other CNS malformations
- rarely occur in isolation
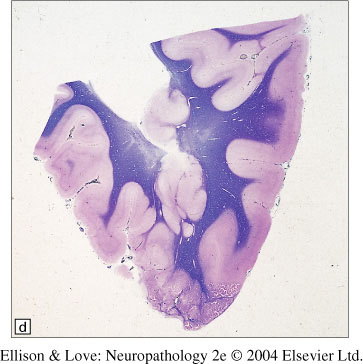
- subcortical band heterotopia - nodular to confluent masses of grey matter between and separated from the ventricular surface and overlying cortex, forming a contiguous band of subcortical whitematter
- also called "double cortex", or "subcortical laminar heterotopia"
- often seen with pachygyria
- usually occur without other malformative lesions
- periventricular heterotopia - nodular masses of grey matter near the ventricular surface
- also called "periventricular nodular heterotopia", "bilateral periventricular nodular heterotopia"
- often seen with other malformations, including microcephaly, agenesis of the corpus callosum, septooptic dysplasia, thanatophoric dysplasia, chromosomal abnormalities, multiorgan syndromes
- FLNA mutation results in relatively pure periventricular heterotopia
Epidemiology
- leptomeningeal heterotopias may be the most common congenital brain malformations, present in 31% infant autopsies
- subcortical band heterotopia is least common
- leptomeningeal - no sex predilection
- subcortical band heterotopia - female predominance
- periventricular heterotopia - genetic form is X-linked, lethal in males. Otherwise no sex predilection
- risk factors are genetic and epigenetic (hemorrhages, hypoxic-ischemic injury, toxins)
Clinical features
- Leptomeningeal heterotopia
- syndromes associated with severe mental retardation and seizures
- associated with seizures due to their cortical location and composition of cortical neurons
- not detectable by current imaging techniques - attractive potential cause for structurally "silent" intractible epilepsy
- if brainstem involved, associated with hydrocephalus
- Subcortical band heterotopia
- females have mild to moderate symptoms
- males have either more normal or more severe symptoms
- most commonly mental retardation and seizures
- seizure types include partial complex seizures with secondary generalization, atypical absence epilepsy with regional or focal onset
- severity of symptoms correlates with extent of heterotopia
- this heterotopic band participates in some normal cognitive processes
- Periventricular heterotopia
- females with X-linked form can have epilepsy and mild mental retardation
- epilepsy often begins in second decade of life, and drug resistant
- partial complex and atypical absence are common forms
- males may have more severe symptoms, with associated cortical dysplasia
- FLNA mutations also may have small corpus callosum, mild cerebellar anomaly, patent ductus arteriosus, coagulopathy, early strokes, digit anomalies
- one recessive form associated with developmental delay
- Heterotopia can be detected by T1 weighted MRI, except leptomeningeal heterotopia, which is generally undetected. Diffuse leptomeningeal heterotopia may be seen as thickening of the leptomeninges or obliteration of the subpial/subarachnoid space
Macroscopy/Histopathology
- cellular composition is normal, including pyramidal (glutaminergic) and non-pyramidal (GABAergic) neurons randomly oriented without obvious lamination
- elements of normal cortex including astrocytes, oligodendrocytes, vessels, neuropil, axons, and scant myelin can be seen
- abnormal features of apoptosis and mineralization typically not seen
- leptomeningeal heterotopia
- usually only seen microscopically, except associated with type II lissencephaly, where irregularities on the brain surface can be appreciated
- focal protrusion of glioneuronal tissue from the surface of the brain onto the leptomeninges and subarachoid space
- connected to the underlying marginal zone via bridges of glioneuronal tissue
- often polypoid with central blood vessel and core of radially oriented fibers
- underlying cortex can have cortical dysplasia, and leptomeninges fibrotic
- GFAP and synaptophysin will be positive in the leptomeninges
- ultrastructural studies suggest a defect in the glia limitans in leptomeningeal heterotopia associated with Fukuyama congenital muscular dystrophy

- Subcortical band heterotopia
- morphology can vary from curvilinear ribbons to nodules
- can be unilateral/bilateral, diffuse/focal
- regional differences in band thickness, possibly associated with the genotype
- appearance of multiple conglomerated nodules, with radially oriented axes, separated by myelinated tracts
- nodules may merge with subcortical grey nuclei or overlying cortex
- cortical surface can appear normal or pachygyric (combined SBH-pachygyria)
- no distinctive histopathology
- cortex overlying it is normal, except in SBH-pachygyria
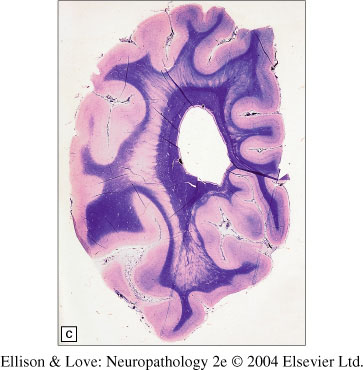
- Periventricular heterotopia
- variable in extent, from isolated single nodules to confluent bilateral lesions
- can distort the lateral ventricles
- can be located away from the ependymal surface
- most commonly near the angle of the lateral ventricles around the temporal horns
- in neonates, can be indistinct due to paucity of myelination
- cortex over this type of heterotopia often shows some abnormalities, commonly polymicrogyria
- nodules can show rudimentary lamination, with neuronal size changes consistent with age
- multiple neuronal types involved, with slight of absent GFAP immunoreactivity
- decreased NMDA receptor in the nodules and overlying dysplastic cortex
- disrupted GFAP positive fibers and CD68 positive macrophages can surround the nodules from epigenetic insults
Genetics
- Leptomeningeal heterotopia
- genetic syndromes include trisomy 13, holoprosencephaly, type II lissencephaly
- genes for type II lissencephaly have been found tht are autosomal recessive and also show diffusely abnormal cortical lamination and architechture, diffuse leptomeningeal heterotopia, and congenital muscular dystrophy
- Subcortical band heterotopis
- DCX ("double cortex") gene on chr Xq22.3-q23
- X-linked dominant inheritance
- heterozygous females have subcortical band heterotopia
- anterior bias of heterotopia
- males have lissencephaly
- LIS1 (17p13.3)
- associated with subcortical band heterotopia in males only
- posterior bias of heterotopia
- other possible genetic causes include mutations in chromosome 9p
- Periventricular heterotopia
- complex genetics due to large number of syndromes that include periventricular heterotopia
- FLNA mutation - X-linked dominant inheritance with prenatal male lethality
- can be sporadic or inherited
- associated with multiorgan syndromes including: otopalatodigital syndrome type 1 and 2, frontometaphyseal dysplasia, Melnick-Needles syndrome
Experimental models
Experimental mouse models have helped to better understand the mechanisms behind heterotopia. For instance, in leptomeningeal heterotopia, it has been shown in mice that there is radial glial-guided migration of neurons to the ectopic tissue, with possibly defects in the preplate. As well, radiation induced damage of the radial glia in mice can cause periventricular heterotopia, suggesting that disruption of radial glial-guided migration may contribute to this result.
Pathogenesis
- The mosaic phenotypes of subcortical band heterotopia and periventricular heterotopia can be explained by X-inactivation in heterozygous females and in germline and somatic mosaicism in the autosomal forms.
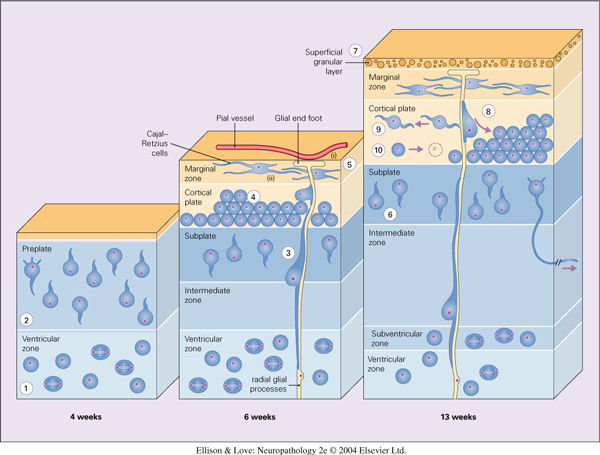
- Defects in radial glial-guided neuronal migration
- radial migration involves (1) onset of migration out of the germinal matrix, (2) ongoing migration through the future white matter (intermediate zone), (3) penetration through the subplate, (4) progression through the cortical plate, and (5) stopping at the marginal zone and pial-glial border.
- periventricular heterotopia involves defect in 1
- subcortical band heterotopia involves defect in 2
- leptomeningeal heterotopia involves defect in 5
- defects in 3 and 4 would result in lamination defects and cortical dysplasias
- Protein glycosylation in leptomeningeal heterotopia
- protein glycosylation defects shared with type II lissencephalies
- potential roles for proteins in the proteoglycan-rich marginal zone and subplate, the glia limitans, or the pial basement membrane
- microtubule dynamics in subcortical band heterotopia
- DCX gene product doublecortin is found in the leading edge of processes to stimlate mitrotubule proliferation
- Lis1 interacts with components of the centrosome important for microtubule dependent translocation of the nucleus
- other cytoskeletal abnormalities in periventricular heterotopia
- FLNA produces Filamin A which is an actin-binding protein that anchors neuronal migration related proteins to the actin cytoskeleton
- other potential molecular mechanisms also have been suggested, including a role for blood vessels in leptomeningeal and periventricular heterotopia
- epigenetic mechanisms are likely quite common in periventricular and leptomeningeal heterotopia. These include:
- germinal matrix or subpial hemorrhages
- periventricular leukomalacia
- hypoxic-ischemic, physical, radiation, chemical, thermal-induced injury